WhakamĀrama Āpitihanga
Hei whakaahua i te putanga mai o te ua kawa:
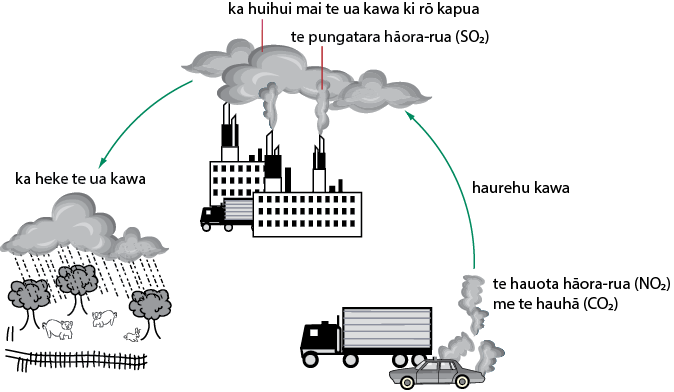
Ka puta te ua kawa i te tauhohenga o ētahi haurehu me te wairehu e puritia ana ki rō kapua. Ko ngā haurehu matua nei ko te pungatara hāora-rua (SO2) me te hauota hāora-rua (NO2). He mea tuku ēnei haurehu ki te kōhauhau mā te tahu koranehe. Ko te waikawa pungatara (H2SO4) me te waikawa hauota (HNO3) te hua o ēnei tauhohenga matū. Kia rahi rawa ēnei matū ki rō kapua, kātahi ka heke te ua kawa ki te whenua. Nā te mahi a te ua kawa, ka pūnguru ētahi rawa hanga whare, ka māngeongeo te kiri o te tangata, ka kawa hoki ngā awa me ngā roto. I ētahi wā, ka mate ngā tipu.
Acid rain results from the reaction of some gases such as sulphur dioxide(SO2) and nitrogen dioxide(NO2) with water vapour in clouds. These gases are released into the atmosphere by the burning of fossil fuels. The reaction of these gases with water vapour produces sulphuric acid(H2SO4) and nitric acid(HNO3). When these chemicals reach significant amounts, acid rain falls to the Earth and can cause corrosion to some building materials, irritation to the skin, pollution to streams and waterways, and sometimes can destroy crops and forests.
ua - rain
kawa (waikawa) - acid